Synthesis is the application of chemical processes for the purpose of forming specific chemical products. Traditional multistep synthesis uses a variety of solvents and reagents; elevates temperature and/or pressure; requires purification after every step; and usually creates a yield far below 100%, thus generating substantial waste. A traditional synthesis procedure, then, generally results in heavy costs (both material and environmental) associated with materials and energy inputs, waste disposal, and regulatory compliance.
Non-Covalent Derivatization (NCD) is an alternative to traditional synthesis. Rather than traditional techniques that modify the properties of a target compound by covalently attaching to it various functional groups, NCD temporarily changes the properties of the starting material by intentionally directing the intermolecular interactions. NCDs combine the starting material with an auxiliary material: they are typically formed in one quick step under ambient conditions by solventless grinding of the reactants. Both reactants are fully incorporated into the product; thus, virtually no waste is generated and no purification of the product is required.
As it is in the case of the traditional, “covalent” derivatization, the resulting derivative has chemical and physical properties different from those of the starting material. The NCD is a compound which in solid state has properties and behaviors entirely different from those of either of the two parent compounds. The properties can be fine-tuned by changing both the ratio of target to auxiliary and the identity of the auxiliary compound. By selecting the optimal auxiliary component of the construct, the construct will disintegrate appropriately, delivering the target compound to the target site. After dissociation, the NCD releases both parent compounds, allowing the target compound to perform its function.The auxiliary component is not being consumed and can be collected for further reuse, if desired.
INDUSTRIAL APPLICATION OF NCD: POLAROID INSTANT PHOTOGRAPHY
Polaroid Instant photography relies on hydroquinone as a developer participating in the photoinduced redox chemistry of silver ions.
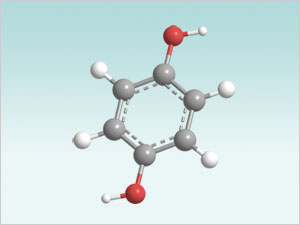
Hydroquinone Molecule
Polaroid Instant photography relies on a developer that must have both shelf stability and exceptional performance. These conflicting requirements are satisfied by a hydroquinone system, which is stable: it is insoluble and immobile at neutral pH, but it rapidly becomes soluble and mobile when its pH is raised to a certain value. Two main problems have to be overcome to use such a system: autoxidation, oxidation of hydroquinone with vagrant oxygen present in the system prior to reaction with silver ions, and premature diffusion, undesired mobility of hydroquinone at neutral pH.
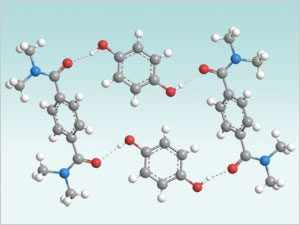
Non-Covalent Derivatization provides the answer to this problem. Cocrystallization of hydroquinone with bis-(N,N-dialkyl) terephthalamides renders hydroquinone stable (with respect to oxidation), less water-soluble, and relatively immobile at neutral pH. At elevated pH, the hydroquinone becomes deprotonated, which disrupts the networks of hydrogen bonds maintaining the structural integrity of the cocrystals, and the hydroquinone is released. Varying the terephthalamide complexing agents and hydroquinone to terephthalamide ratios affects the weak interactions between the molecules in the cocrystal, which results in a variety of crystal structures exhibiting different motifs of hydrogen bond networks, thus allowing fine control over the hydroquinone properties.
Non-Covalent Derivatization: Terephthalamide (TPA) – Hydroquinone (HQ) motifs. Different crystal structures (motifs) enable control of hydroquinone release properties allowing fine tuning of product performance.
The environmental and economic benefits of this new approach to hydroquinone derivatization are significant. Noncovalent derivatization of hydroquinone is performed in one step by solventless grinding, aqueous attriting, or simply adding terephthalamide to the recrystallization liquor of crude hydroquinone, thus avoiding the need for organic solvents, purification procedures, energy inputs, and waste disposal associated with traditional covalent derivatization.
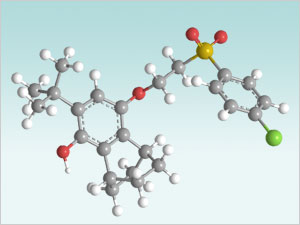
Traditional Approach: Covalent derivatization of hydroquinone molecule, requiring a multi-step synthetic procedure requiring several solvents, high energy inputs, and hazardous reagents.